Oncolytics Biotech, trading under the ticker (ONCY, Financial), has successfully met Nasdaq's listing standards, as confirmed by the Nasdaq Stock Market's Listing Qualifications Department. A formal notification issued on July 22 acknowledged that the company complies with the minimum bid price requirement specified in Listing Rule 5550(a)(2). Consequently, Oncolytics continues to be listed on the Nasdaq Capital Market, maintaining its presence under the same ticker symbol, ONCY.
Wall Street Analysts Forecast
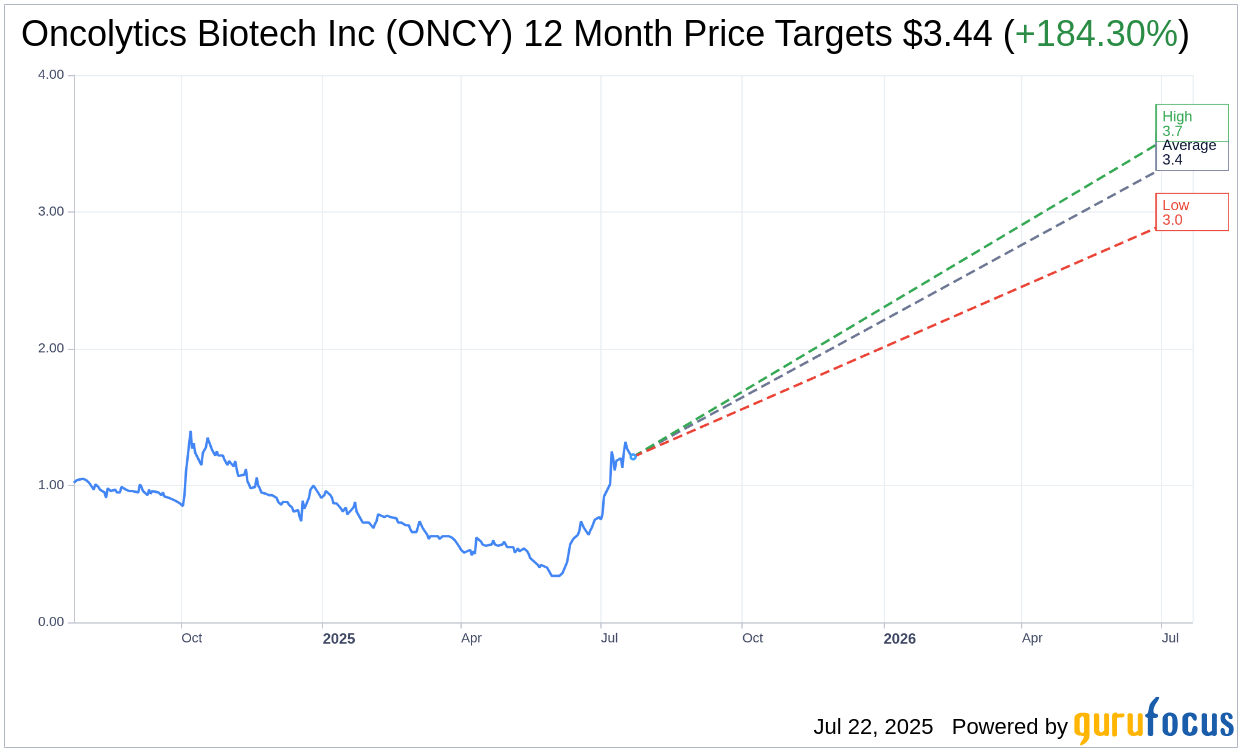
Based on the one-year price targets offered by 3 analysts, the average target price for Oncolytics Biotech Inc (ONCY, Financial) is $3.44 with a high estimate of $3.65 and a low estimate of $3.00. The average target implies an upside of 184.30% from the current price of $1.21. More detailed estimate data can be found on the Oncolytics Biotech Inc (ONCY) Forecast page.
Based on the consensus recommendation from 5 brokerage firms, Oncolytics Biotech Inc's (ONCY, Financial) average brokerage recommendation is currently 2.2, indicating "Outperform" status. The rating scale ranges from 1 to 5, where 1 signifies Strong Buy, and 5 denotes Sell.
ONCY Key Business Developments
Release Date: May 14, 2025
- Cash and Cash Equivalents: $15.3 million as of March 31, 2025.
- Net Cash Operating Activities: $6.5 million for the quarter, down from $7.5 million in the same period last year.
- General and Administrative Expenses: $3 million for the first quarter, consistent with the prior year.
- Research and Development Expenses: $4.1 million, down from $5.7 million in Q1 of 2024.
- Net Loss: $6.7 million or $0.08 per basic and diluted share, compared to $6.9 million or $0.09 per share in Q1 of 2024.
- Share Purchase Agreement: USD20 million agreement with Alumni Capital to extend financial runway.
For the complete transcript of the earnings call, please refer to the full earnings call transcript.
Positive Points
- Oncolytics Biotech Inc (ONCY, Financial) reported promising clinical data for pelareorep, showing a 33% objective response rate in relapsed anal carcinoma, including a complete response lasting over 15 months.
- The company is actively enrolling patients in the GOBLET Cohort 5 study for metastatic pancreatic cancer, supported by a $5 million grant from the Pancreatic Cancer Action Network.
- Pelareorep demonstrated a favorable safety profile across multiple studies, allowing for combination with various chemotherapies and checkpoint inhibitors.
- The company has secured a USD20 million share purchase agreement with Alumni Capital, providing financial flexibility and extending the runway through key milestones.
- Oncolytics Biotech Inc (ONCY) is exploring multiple registrational opportunities for pelareorep in breast cancer, supported by positive data from two randomized Phase 2 studies showing significant clinical benefits.
Negative Points
- The company is still in the process of searching for a new CEO, which may impact strategic decision-making and leadership continuity.
- Despite promising data, regulatory approval for pelareorep in anal carcinoma is not guaranteed and the commercial opportunity is smaller compared to other indications.
- Research and development expenses decreased due to reduced manufacturing and clinical trial costs, which could indicate a slowdown in certain development activities.
- The net loss for the quarter was $6.7 million, reflecting ongoing financial challenges despite a slight improvement from the previous year.
- There have been no recent interactions with the FDA regarding the pancreatic cancer program, which could delay potential registrational pathways.
